
The Power of Automation in Data Migration
Automation is a practical solution to speed up and optimize the entire data migration process
Cell and gene therapies (CGT) have revolutionized the field of medicine, offering innovative treatment options for previously incurable diseases. Congruent to “traditional” medicines, CGT require a functioning data management supporting the patient’s health. This article explores the importance of MDM in the context of CGT and presents an approach for effective data management in this rapidly advancing field.
According to the U.S. Food & Drug Administration (FDA), cell therapies encompass, for example, immunotherapies or cancer vaccines, gene therapies aim to modify a patient sample with following reinjection (FDA, 2023a). Focusing on various products, nearly all relevant market players are involved in the production. The following 33 cell and gene therapy products are approved on the US market until the end of 2023 (FDA, 2023b).
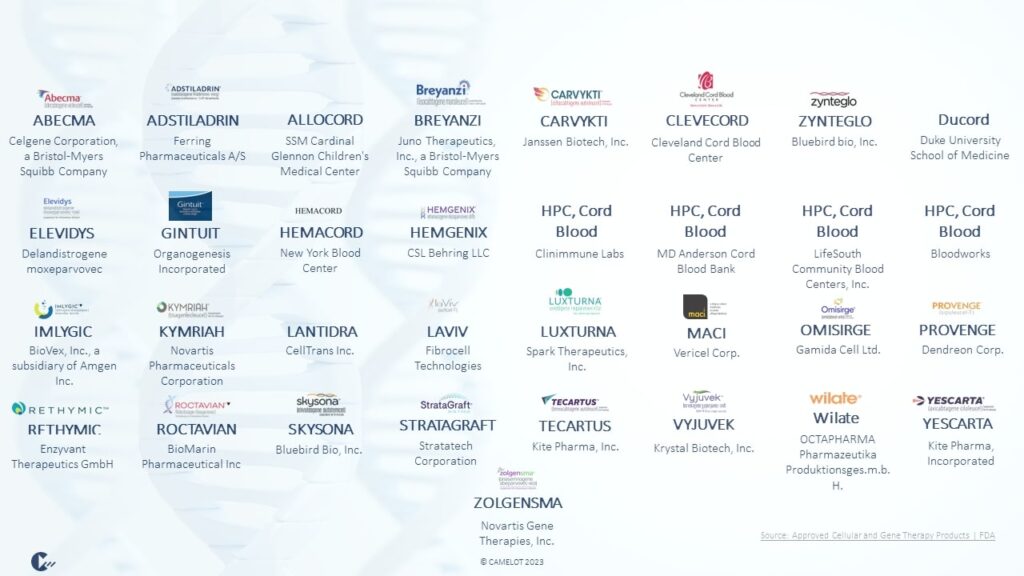
These innovative CGT products require dedicated supply chains because the products have an extraordinarily high value but low volume. Furthermore, the CGT supply chains are highly collaborative with numerous stakeholders, which demands visibility and control to ultimately ensure the patient’s life (Göbel & Mendes, 2022). Taken together, this results in a high level of uncertainty in the CGT environment. Therefore, a must in CGT considerations is Master Data Management (MDM), which plays a critical role in ensuring the accuracy, integrity, and accessibility of central data to make informed decisions.
Master Data Management refers to the core data elements that are shared across an organization, providing a single point of reference, and ensuring consistency throughout different systems and processes. In the context of cell and gene therapies, MDM involves the centralized management of critical data related to patients, treatments, clinical trials, manufacturing processes, and regulatory compliance. We at Camelot developed the heatmap approach to continuously evaluate the maturity of CGT MDM. The heatmap is an evolution of classical data management approaches (Aumann & Jendrossek, 2021), encapsulating additional dimensions and continuously monitoring them. Through monitoring, changing priorities and urgencies are identified, and the color of a dimension changes.
In general, the heatmap dimensions are Governance & Data Readiness, MDM Processes, Data & Integration, User Interface, General Functionalities, and Data Privacy & Compliance. These dimensions are explained briefly in the following:
Governance & Data Readiness
Analyzing this dimension, it is essential to establish the ownership of fields for accountability and accuracy, as it designates individuals responsible for specific data elements. Furthermore, clearly defined roles and responsibilities within a data governance framework streamline decision-making processes and enhance overall data quality. Moreover, the concept of data readiness involves preparing data for use by ensuring its accuracy, consistency, and compliance with established standards. This readiness ensures that data is reliable and meets the needs of users. Besides, a well-structured approach to data load, encompassing data extraction, transformation, and loading processes, is indispensable for maintaining a robust and up-to-date data environment.
Master Data Management Processes
The process dimension involves the creation of new master data entries, ensuring that accurate and relevant information is captured from the outset. The change process is equally crucial, allowing for updates and modifications to existing master data to reflect the evolving needs of the business. In addition, the consideration of the deletion process is important, which necessitates a systematic and controlled approach to remove outdated or redundant data, preventing inaccuracies and minimizing data bloat. Finally, Standard Operating Procedures (SOP) and comprehensive training programs are essential components of master data maintenance, providing guidelines and ensuring that personnel involved in these processes are well-equipped with the necessary knowledge and skills.
Data & Integration
In the CGT MDM environment, the key components of this dimension such as Data Model, Data Dictionary, and adherence to Data Standards. The Data Model serves as the blueprint, outlining the structure and relationships of data entities, and providing a visual representation for better understanding and organization. A comprehensive Data Dictionary acts as a centralized repository, defining data elements, their meanings, and usage, ensuring consistency across the organization. Data Standards play a pivotal role in this by establishing guidelines for data formatting, quality, and integration, fostering interoperability, and facilitating seamless data exchange between systems.
User Interface
The User Interface (UI) is a crucial aspect of CGT systems, shaping the user experience and facilitating effective interaction. First, field conditions within the UI ensure that data input aligns with predefined criteria, promoting accuracy and consistency. Second, system help features provide users with guidance and support, offering context-specific information to enhance usability and reduce errors. Lastly, the inclusion of notes or remarks in the UI allows users to add relevant context or details, fostering communication and collaboration within the system. A well-designed UI, encompassing these elements, not only empowers users to navigate and interact seamlessly but also contributes to overall system efficiency and user satisfaction.
General Functionalities
In CGT MDM, General Functionalities encompass essential features that contribute to overall efficiency and effectiveness. The inclusion of a duplicate check functionality ensures data integrity by preventing the inadvertent creation of redundant information. Mass capabilities streamline processes by allowing users to perform actions on multiple records simultaneously, saving time and enhancing productivity. Data reports provide valuable insights into system performance and trends, enabling informed decision-making.
Data Privacy & Compliance
Ultimately, the dimension Data Privacy & Compliance is particularly meaningful in CGT MDM. Companies respecting Data Privacy & Compliance support regulatory audits and ensure adherence to regulatory standards. To do so, an authorization concept establishes access controls, ensuring that users only interact with data relevant to their roles, safeguarding sensitive information. On top of that, the implementation of an audit trail records and monitors all system activities, providing a comprehensive history that aids in compliance assessments and forensic analyses. Besides, the “4-eyes principle” adds an extra layer of security by requiring a second individual to review and approve critical actions, minimizing the risk of unauthorized or inappropriate activities.
The article underscores the critical role of Master Data Management (MDM) in the context of cell and gene therapies (CGT), emphasizing its importance in ensuring the accuracy, integrity, and accessibility of central data for informed decision-making. The CGT supply chains, described as highly collaborative, demand visibility and control for patient safety, making MDM a crucial consideration. In this article, the heatmap approach with dimensions such as Governance & Data Readiness, MDM Processes, Data & Integration, User Interfaces, General Functionalities, and Data Privacy & Compliance is presented ensuring a qualitative CGT MDM assessment.
The creation of the blog post was supported by Artificial Intelligence.
We would like to thank Robert Rössler for his valuable contribution to this article.

Automation is a practical solution to speed up and optimize the entire data migration process

Discover how you can streamline your financial master data management and reengineer financial processes with SAP MDG Finance.

Whilst it seems straightforward to carry out either a blood transfusion or a data migration, the actuality proves far more complex.

In this blog article, Camelot introduces you to the main principals of Demand-Driven Supply Chain Management.